Review of safety and minimum pricing of nitazoxanide for potential treatment of COVID-19
| Author List |
|---|
| Toby Pepperrell |
| Victoria Pilkington |
| Andrew Owen |
| Junzheng Wang |
| Andrew Hill |
Abstract
Background: Many treatments are being assessed for repurposing to treat coronavirus disease 2019 (COVI D-19). One drug that has shown promising results in vitro is nitazoxanide. Unlike other postulated drugs, nitazoxanide shows a high ratio of maximum plasma concentration (Cmax), after 1 day of 500 mg twice daily (BD), to the concentration required to inhibit 50% replication (EC50) of severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) (Cmax : E C50 roughly equal to 14:1). As such, it is important to investigate the safety of nitazoxanide for further trials. Furthermore, treatments for COVI D-19 should be cheap to promote global access, but prices of many drugs are far higher than the costs of production. We aimed to conduct a review of the safety of nitazoxanide for any prior indication and calculate its minimum costs of production.
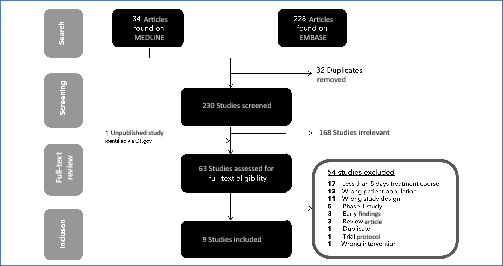
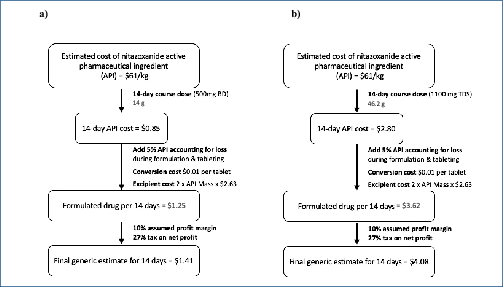
Methods: A review of nitazoxanide clinical research was conducted using EMBASE and MEDLINE databases, supplemented by ClinicalTrials.gov. We searched for phase 2 or 3 randomised controlled trials (RCTs) comparing nitazoxanide with placebo or active control for 5–14 days in participants experiencing acute infections of any kind. Data extracted were grade 1–4 and serious adverse events (AEs). Data were also extracted on gastrointestinal (GI) AEs, as well as hepatorenal and cardiovascular effects. Active pharmaceutical ingredient cost data from 2016 to 2019 were extracted from the Panjiva database and adjusted for 5% loss during production, costs of excipients, formulation, a 10% profit margin and tax. Two dosages, at 500 mg BD and a higher dose of 1100 mg three times daily (TDS), were considered. Our estimated costs were compared with publicly available list prices from a selection of countries.
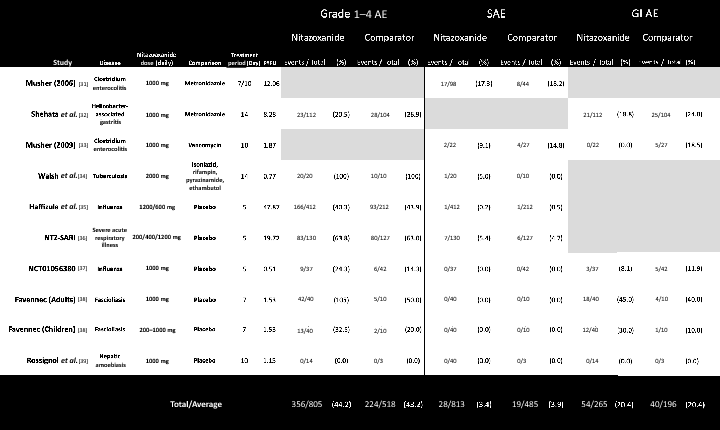
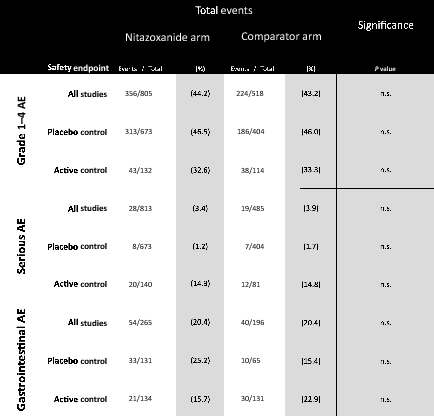
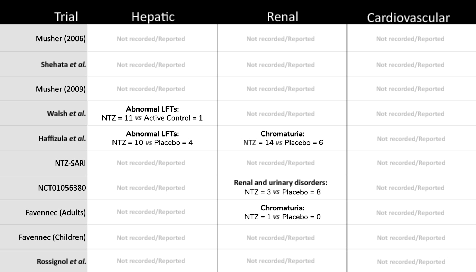
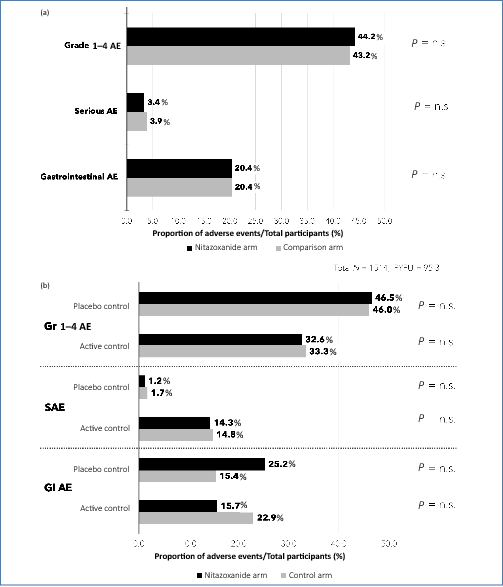
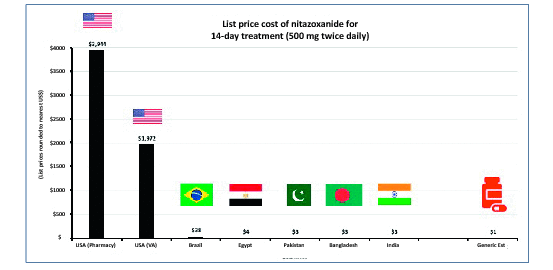
Results: Nine RCTs of nitazoxanide were identified for inclusion. These RCTs accounted for 1514 participants and an estimated 95.3 person-years-of-follow-up. No significant differences were found in any of the AE endpoints assessed, across all trials or on subgroup analyses of active- or placebo-controlled trials. Mild GI AEs increased with dose. No hepatorenal or cardiovascular concerns were raised, but few appropriate metrics were reported. There were no teratogenic concerns, but the evidence base was very limited. Based on a weighted-mean cost of US $61/kg, a 14-day course of treatment with nitazoxanide 500 mg BD would cost $1.41. The daily cost would therefore be $0.10. The same 14-day course could cost $3944 in US commercial pharmacies, and $3 per course in Pakistan, India and Bangladesh. At a higher dose of 1100 mg TDS, our estimated cost was $4.08 per 14-day course, equivalent to $0.29 per day.
Conclusion: Nitazoxanide demonstrates a good safety profile at approved doses. However, further evidence is required regarding hepatorenal and cardiovascular effects, as well as teratogenicity. We estimate that it would be possible to manufacture nitazoxanide as generic for $1.41 for a 14-day treatment course at 500 mg BD, up to $4.08 at 1100 mg TDS. Further trials in COVI D-19 patients should be initiated. If efficacy against SARS-CoV-2 is demonstrated in clinical studies, nitazoxanide may represent a safe and affordable treatment in the ongoing pandemic.
Published
Article Category
COVID-19
Article Type
Reviews
Posted Date
30-04-2020